Lewis structure sf26/6/2023 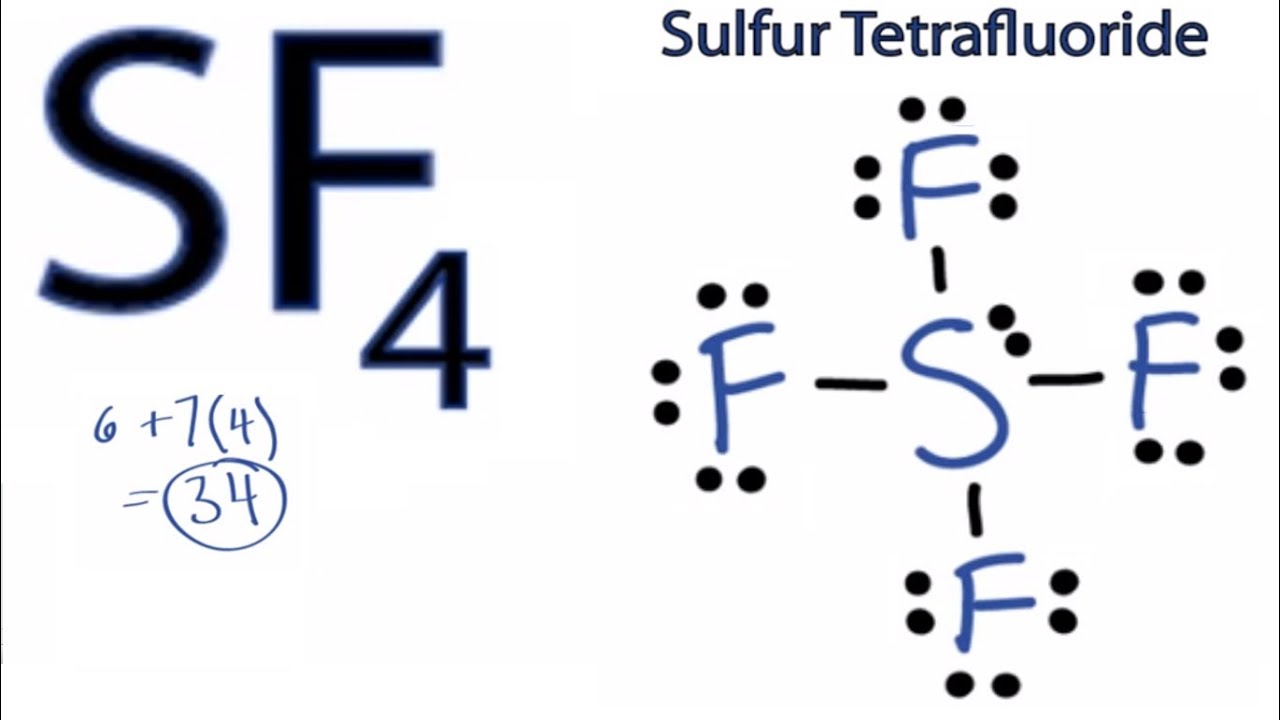
Dream is a little unclear about how to draw Lewis structures. It has a bent geometry with single bonds to each fluorine and two lone pairs on the sulfur. There does exist a neutral sulfur difluoride molecule. 
Assuming the anion exists, it would have 21 electrons, six for sulfur and seven each for the fluorine atoms, plus one more because of its "charge". You seem to have trouble counting electrons. Fluorine has 7 valence electrons, not six. Free radicals tend to be quite reactive because of the single unpaired electron. For one thing, it would be a free radical, meaning that it has an odd number of electrons.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
